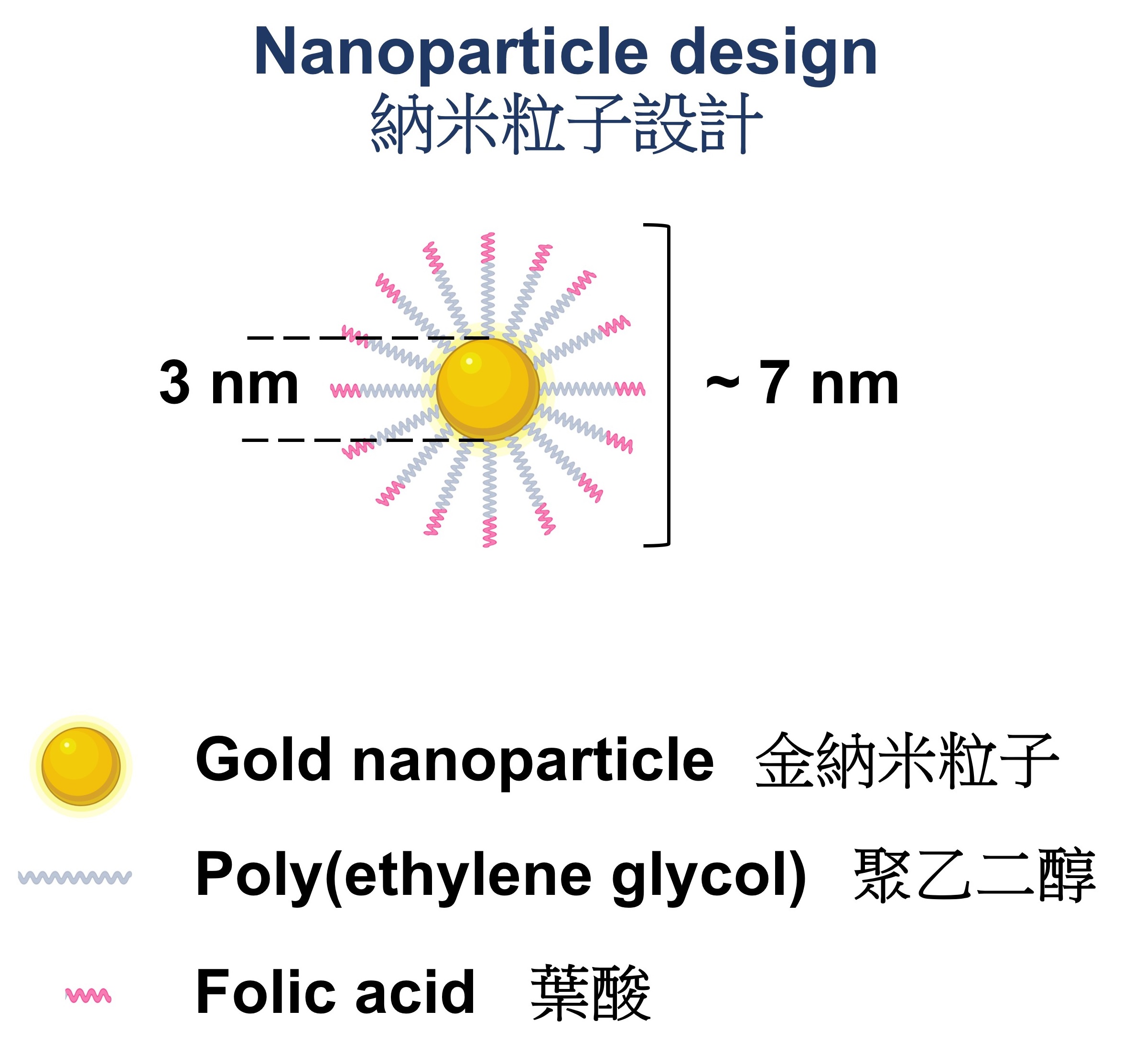
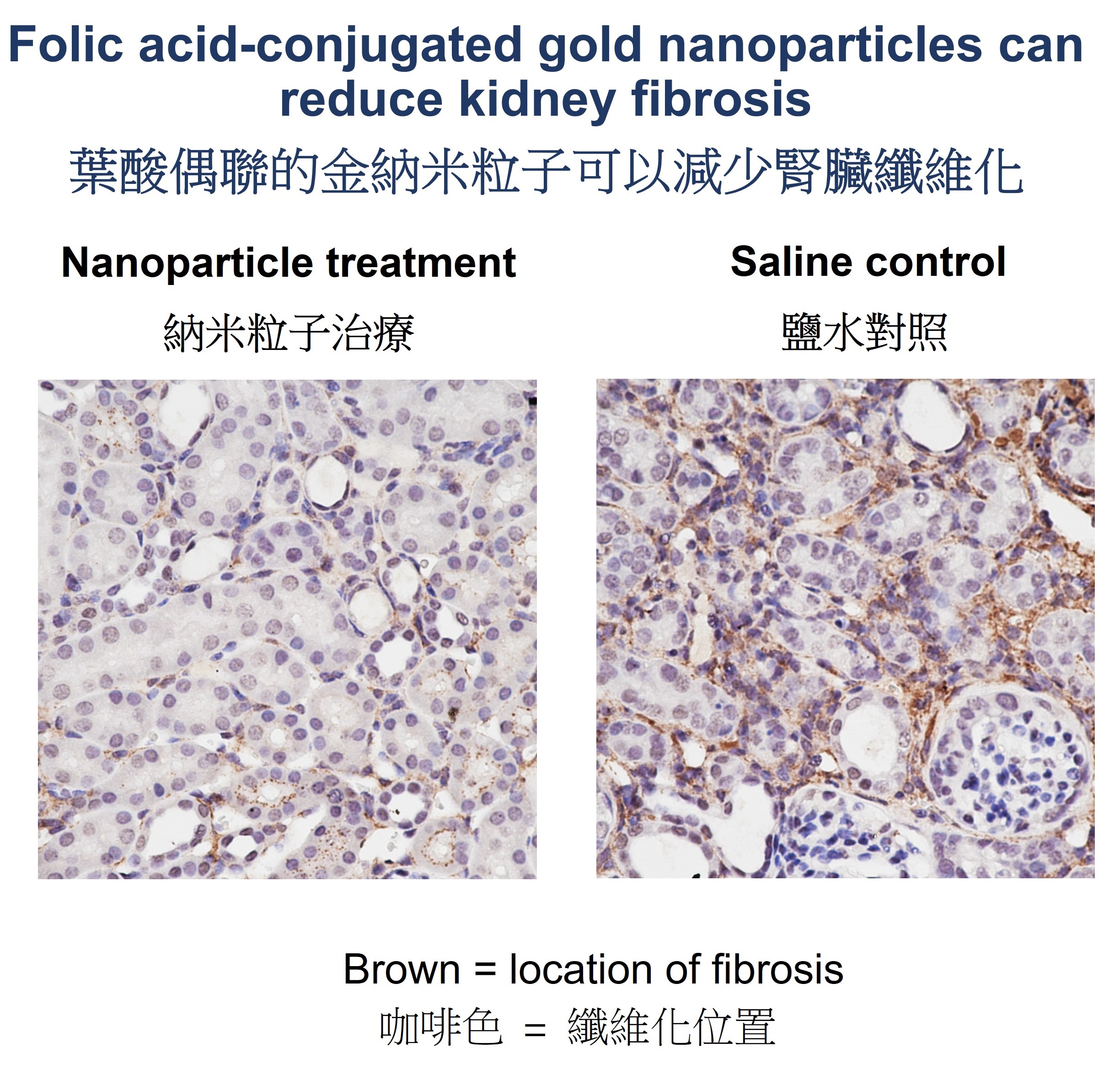
Prolonged kidney fibrosis can lead to chronic kidney disease (CKD) and ultimately kidney failure, which requires dialysis or transplantation. A research team led by Professor Jonathan Choi Chung-hang, Associate Professor in the Department of Biomedical Engineering at The Chinese University of Hong Kong (CUHK)’s Faculty of Engineering, has developed a folic-acid conjugated gold nanoparticle that may offer a safe, effective treatment for kidney fibrosis. The research has recently been published in the prestigious journal The Proceedings of the National Academy of Sciences (PNAS). Currently, there are no specific treatments for kidney fibrosis, a major pathological hallmark of CKD. Medications are available to control blood pressure and reduce the rate of disease progression, but they are not entirely effective, and adverse effects such as hypotension and hyperkalemia are common. Nanomedicines hold great potential to treat CKD, but delivering them to renal tubules where fibrosis occurs is still challenging. Bypassing the kidney delivery bottleneck with nanotechnology Nanoparticles are promising drug carriers to the kidneys because their delivery can be tailored by adjusting the nanoparticles’ physicochemical properties. Many existing kidney nanomedicines employ large nanoparticles (larger than 100 nm in diameter) to carry sufficient drugs and targeting ligands, but they need to disassemble unpredictably in the bloodstream to cross the glomerular filtration barrier (with a typical upper size limit smaller than 10 nm) before reaching the renal tubules where fibrosis occurs. Conversely, ultra-small nanoparticles (smaller than 5 nm) can pass through the glomerular filtration barrier, but they are often rapidly cleared from the body via urine, with limited retention in the kidney for therapeutic purposes. To bypass this delivery bottleneck to the kidneys, Professor Choi’s team has invented a design to transport nanoparticles into the renal tubules of fibrotic kidneys. This ~7-nm particle can cross the glomerular filtration barrier without rapid urine clearance. To the surface of a 3-nm gold nanoparticle, the researchers have attached about 32 molecules of folic acids to form a folic-acid conjugated gold nanoparticle with a total size of around 7 nm. The conjugated folic acids can bind to the folate receptors on kidney tubules inside the fibrotic kidney, enhancing the intravenous delivery of the gold nanoparticles to the kidney. Professor Szeto Cheuk-chun in the Department of Medicine and Therapeutics at CUHK’s Faculty of Medicine (CU Medicine) said, “We discovered the localisation of folate receptors on some tubules in both mouse kidneys and biopsy samples from CKD patients.” Dr Cecilia Chan Ka-wing, the first author of the publication and PhD graduate in CU Medicine’s Department of Surgery, added, “Our nanoparticle can preferentially accumulate in the fibrotic kidney, while leaving the other healthy kidney unaffected.” Kidney fibrosis can be treated with a single injection of nanoparticles Past CKD nanomedicines were mostly preventive. The researchers have found that a single injection of folic acid-conjugated gold nanoparticles after the disease has become established was sufficient to reduce tissue degeneration, treat kidney fibrosis, and inhibit genes related to the extracellular matrix. Professor James Lau Yun-wong from CU Medicine’s Department of Surgery stated, “This point is of clinical significance; as early development of CKD is asymptomatic, patients are mostly diagnosed at a later stage.” The nanoparticles can be eventually cleared from the body via urine and faeces, and no pronounced toxicity was observed seven days after injection. Professor Choi concluded, “Gold nanoparticles smaller than 10 nm are self-therapeutic for kidney fibrosis. They can inhibit p38α MAPK, an enzyme that contributes to the development of CKD, without the aid of any chemical or biological drugs. We hope to continue our collaboration with CU Medicine and to validate the safety and efficacy of gold nanoparticles in humans. Ultimately, we hope to offer a safe, effective gold nanomedicine for patients with kidney disease.”
|
|